2.5 ENZYMES:
Essential Idea: Experimental design – accurate, quantitative measurements in enzyme experiments require replicates to ensure reliability.
Enzymes Understanding's:
- Enzymes have an active site to which specific substrates bind.
- Enzyme catalysis involves molecular motion and the collision of substrates with the active site.
- Temperature, pH and substrate concentration affect the rate of activity of enzymes.
- Enzymes can be denatured.
- Immobilized enzymes are widely used in industry.
Enzymes Application's:
- Methods of production of lactose-free milk and its advantages.
Enzymes Skill's:
- Design of experiments to test the effect of temperature, pH and substrate concentration on the activity of enzymes.
- Experimental investigation of a factor affecting enzyme activity.
Active Sites & Enzymes:
- Enzymes are globular proteins that work as catalysts- they speed up chemical reactions without being altered themselves.
- Enzymes are often called biological catalysts because they are made by living cells and speed up biochemical reactions. The substances that enzymes convert into products in these reactions are called substrates.
- Enzymes are found in all living cells and are also secreted by some cells to work outside. Living organisms produce many different enzymes- literally thousands of them.
- Many different enzymes are needed, as enzymes only catalyze one biochemical reaction and thousands of reactions take place in cells, nearly all of which need to be catalyzed.
- This property is called enzyme-substrate specificity.
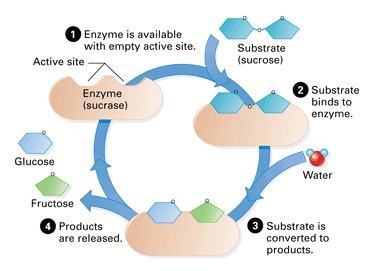
- In order to explain enzyme-substrate specificity, we must look at the mechanism by which enzymes speed up reactions. This involves the substrate, or substrates binding to a special region on the surface of the enzyme called the active site.
- The shape and chemical properties of the active site and the substrate match each other. This allows the substrate to bind, but not other substances. Substrates are covered into products while they are bound to the active sire and the products are then released, freeing the active site to catalyze another reaction.
Enzyme Activity:
Enzyme activity is the catalysis of a reaction by an enzyme and there are three stages:
1. The substrate binds to the active site of an enzyme. Some enzymes have two substrates that bind to different parts of the active site.
2. While the substances are bound to the active site they change into different chemical substances, which are the products of reaction.
3. The products separate from the active site, leaving it vacant for substrates to bind again.
- A substrate molecule can only bind to the active site if it moves very close to it. The coming together of a substrate molecule and an active site is known as a collision. Which suggests a high velocity impact.
- With most reactions the substrates are dissolved in water around the enzyme. Because water is a liquid state, its molecule and all the particles dissolved in it are in contact with each other and are in continual motion. Each particle can move separately. The direction of movement repeatedly changes and is random, which is the bases of diffusion in liquids,
- Both substrates and enzymes with active sites are able to move, though most substrates molecules are smaller than the enzyme so their movement is faster.
- Collisions between substrate molecules and the active site occur because of random movements of both substrate and enzyme.
- Successful collisions are ones in which the substrate and active site are correctly aligned to allow binding to take place.
Factors Affecting Enzyme Activity:
- In liquids, the particles are in continual random motion. When a liquid is heated, the particles in it are not given more kinetic energy. Both enzyme and substrate molecules therefore move around faster at higher temperatures and the chance of a substrate molecule colliding with the active site of the enzyme is increased. Enzyme activity therefore increases.
- When enzymes are heated, bonds in the enzyme vibrate more and the chance of the bonds breaking increases. When bonds in the enzyme break, the structure of the enzyme changes, including the active site. The change is permanent and is called denaturation. When an enzyme molecule has been denatured, it is no longer able to catalyze reactions. As more and more enzyme molecules in a solution become denatured, enzyme activity falls.
Enzymes are sensitive to pH-
- The pH scale is used to measure the acidity or alkalinity of a solution. The lower the pH, the more acid or the less alkaline a solution is. Acidity is due to the presence of hydrogen ions, so the lower the pH, the higher the hydrogen ion concentration. The pH scale is logarithmic.
- A solution at pH 7 is neutral. A solution at pH 6 is slightly acidic; pH 5 is ten times more acidic than pH 6, pH 4 is one hundred times more acidic than pH 6, and so on.
- Most enzymes have an optimum pH at which their activity is highest. If the pH is increased or decreased from the optimum, enzyme activity decreases and eventually stops altogether,
- Enzymes do not all have the same pH optimum - in fact, there is a wide range. This reflects the wide range of pH environments in which enzymes work.
Enzyme activity is affected by substrate concentration-
- Enzymes cannot catalyze reactions until the substrate binds to the active site. This happens because of the random movements of molecules in liquids that result in collisions between substrates and active sites. If the concentration of substrates is increased, substrate- active site collisions will take place more frequently and the rate at which the enzyme catalyzes its reaction increases.
- After the binding of a substrate to an active site, the active site is occupied and unavailable to other substrate molecules until products have been formed and released from the active site.
Denaturation:
- Enzymes are proteins, and like other proteins their structure can be irreversibly altered by certain conditions. This process is denaturation and both high temperatures and either high or low pH can cause it.
- When an enzyme has been denatured, the active site is altered so the substrate can no longer bind, or if it binds the reaction that the enzyme normally catalyzes does not occur.
- In many cases denaturation causes enzymes that were dissolved in water to become insoluble and form a precipitate.
Immobilized Enzymes:
- In 1897 the Buchner brothers, Hans and Eduard, showed that an extract of yeast, containing no yeast cells, would convert sucrose into alcohol. The door was opened to the use of enzymes to catalyze chemical processes outside living cells.
- Louis Pasteur had claimed that fermentation of sugars to alcohol could only occur if living cells were present. This was part of the theory of vitalism, which stated that substances in animals and plants can only be made under the influence of a "vital spirit" or "vital force",
- More than 500 enzymes now have commercial uses.
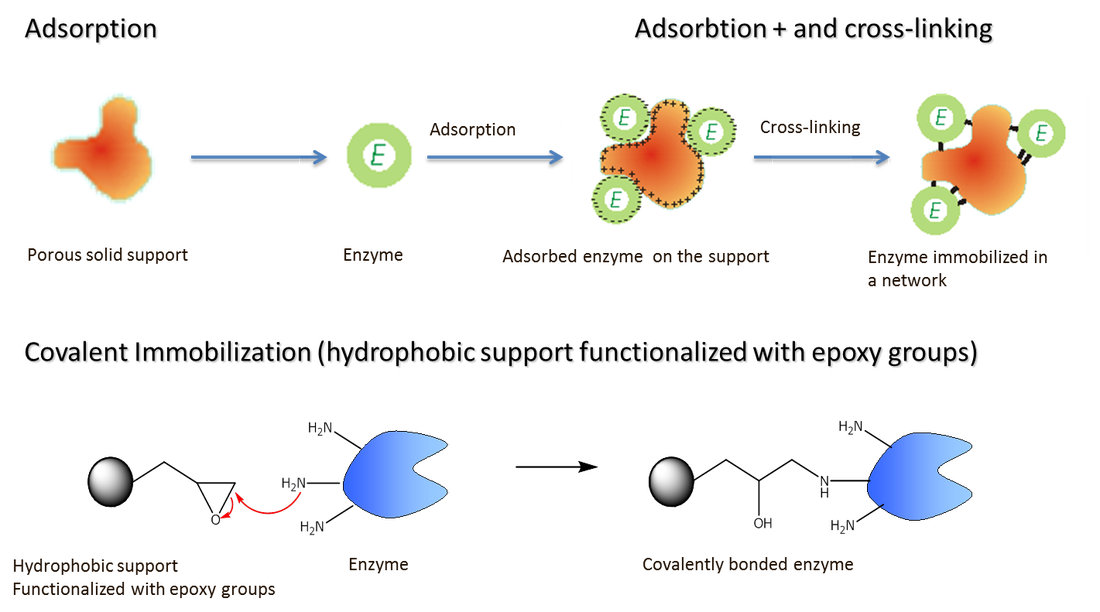
- The enzymes used in industry are usually immobilized. This is attachment of the enzymes to another material into aggregations, so that movement of the enzyme is restricted. There are many ways of doing this, including attaching the enzymes to a glass surface, trapping them in an alginate gel, or bonding them together to form enzyme aggregates of up to 0.1 mm diameter.
Enzyme immobilization advantages-
- The enzyme can easily be separated from the products of the reaction, stopping the reaction at the ideal time and preventing contamination of the products.
- After being retrieved from the reaction mixture the enzyme ma be recycled, giving useful cost saving, especially as many enzymes are very expensive.
- Immobilization increases the stability of enzymes to changes in temperature and pH, reducing the rate at which they are degraded and have to be replaced,
- Substrates can be exposed to higher enzyme concentrations than with dissolved enzymes, speeding up reaction rates.
Enzymes Understanding's:
- Enzymes have an active site to which specific substrates bind.
- Enzyme catalysis involves molecular motion and the collision of substrates with the active site.
- Temperature, pH and substrate concentration affect the rate of activity of enzymes.
- Enzymes can be denatured.
- Immobilized enzymes are widely used in industry.
Enzymes Application's:
- Methods of production of lactose-free milk and its advantages.
Enzymes Skill's:
- Design of experiments to test the effect of temperature, pH and substrate concentration on the activity of enzymes.
- Experimental investigation of a factor affecting enzyme activity.
Active Sites & Enzymes:
- Enzymes are globular proteins that work as catalysts- they speed up chemical reactions without being altered themselves.
- Enzymes are often called biological catalysts because they are made by living cells and speed up biochemical reactions. The substances that enzymes convert into products in these reactions are called substrates.
- Enzymes are found in all living cells and are also secreted by some cells to work outside. Living organisms produce many different enzymes- literally thousands of them.
- Many different enzymes are needed, as enzymes only catalyze one biochemical reaction and thousands of reactions take place in cells, nearly all of which need to be catalyzed.
- This property is called enzyme-substrate specificity.
- In order to explain enzyme-substrate specificity, we must look at the mechanism by which enzymes speed up reactions. This involves the substrate, or substrates binding to a special region on the surface of the enzyme called the active site.
- The shape and chemical properties of the active site and the substrate match each other. This allows the substrate to bind, but not other substances. Substrates are covered into products while they are bound to the active sire and the products are then released, freeing the active site to catalyze another reaction.
Enzyme Activity:
Enzyme activity is the catalysis of a reaction by an enzyme and there are three stages:
1. The substrate binds to the active site of an enzyme. Some enzymes have two substrates that bind to different parts of the active site.
2. While the substances are bound to the active site they change into different chemical substances, which are the products of reaction.
3. The products separate from the active site, leaving it vacant for substrates to bind again.
- A substrate molecule can only bind to the active site if it moves very close to it. The coming together of a substrate molecule and an active site is known as a collision. Which suggests a high velocity impact.
- With most reactions the substrates are dissolved in water around the enzyme. Because water is a liquid state, its molecule and all the particles dissolved in it are in contact with each other and are in continual motion. Each particle can move separately. The direction of movement repeatedly changes and is random, which is the bases of diffusion in liquids,
- Both substrates and enzymes with active sites are able to move, though most substrates molecules are smaller than the enzyme so their movement is faster.
- Collisions between substrate molecules and the active site occur because of random movements of both substrate and enzyme.
- Successful collisions are ones in which the substrate and active site are correctly aligned to allow binding to take place.
Factors Affecting Enzyme Activity:
- In liquids, the particles are in continual random motion. When a liquid is heated, the particles in it are not given more kinetic energy. Both enzyme and substrate molecules therefore move around faster at higher temperatures and the chance of a substrate molecule colliding with the active site of the enzyme is increased. Enzyme activity therefore increases.
- When enzymes are heated, bonds in the enzyme vibrate more and the chance of the bonds breaking increases. When bonds in the enzyme break, the structure of the enzyme changes, including the active site. The change is permanent and is called denaturation. When an enzyme molecule has been denatured, it is no longer able to catalyze reactions. As more and more enzyme molecules in a solution become denatured, enzyme activity falls.
Enzymes are sensitive to pH-
- The pH scale is used to measure the acidity or alkalinity of a solution. The lower the pH, the more acid or the less alkaline a solution is. Acidity is due to the presence of hydrogen ions, so the lower the pH, the higher the hydrogen ion concentration. The pH scale is logarithmic.
- A solution at pH 7 is neutral. A solution at pH 6 is slightly acidic; pH 5 is ten times more acidic than pH 6, pH 4 is one hundred times more acidic than pH 6, and so on.
- Most enzymes have an optimum pH at which their activity is highest. If the pH is increased or decreased from the optimum, enzyme activity decreases and eventually stops altogether,
- Enzymes do not all have the same pH optimum - in fact, there is a wide range. This reflects the wide range of pH environments in which enzymes work.
Enzyme activity is affected by substrate concentration-
- Enzymes cannot catalyze reactions until the substrate binds to the active site. This happens because of the random movements of molecules in liquids that result in collisions between substrates and active sites. If the concentration of substrates is increased, substrate- active site collisions will take place more frequently and the rate at which the enzyme catalyzes its reaction increases.
- After the binding of a substrate to an active site, the active site is occupied and unavailable to other substrate molecules until products have been formed and released from the active site.
Denaturation:
- Enzymes are proteins, and like other proteins their structure can be irreversibly altered by certain conditions. This process is denaturation and both high temperatures and either high or low pH can cause it.
- When an enzyme has been denatured, the active site is altered so the substrate can no longer bind, or if it binds the reaction that the enzyme normally catalyzes does not occur.
- In many cases denaturation causes enzymes that were dissolved in water to become insoluble and form a precipitate.
Immobilized Enzymes:
- In 1897 the Buchner brothers, Hans and Eduard, showed that an extract of yeast, containing no yeast cells, would convert sucrose into alcohol. The door was opened to the use of enzymes to catalyze chemical processes outside living cells.
- Louis Pasteur had claimed that fermentation of sugars to alcohol could only occur if living cells were present. This was part of the theory of vitalism, which stated that substances in animals and plants can only be made under the influence of a "vital spirit" or "vital force",
- More than 500 enzymes now have commercial uses.
- The enzymes used in industry are usually immobilized. This is attachment of the enzymes to another material into aggregations, so that movement of the enzyme is restricted. There are many ways of doing this, including attaching the enzymes to a glass surface, trapping them in an alginate gel, or bonding them together to form enzyme aggregates of up to 0.1 mm diameter.
Enzyme immobilization advantages-
- The enzyme can easily be separated from the products of the reaction, stopping the reaction at the ideal time and preventing contamination of the products.
- After being retrieved from the reaction mixture the enzyme ma be recycled, giving useful cost saving, especially as many enzymes are very expensive.
- Immobilization increases the stability of enzymes to changes in temperature and pH, reducing the rate at which they are degraded and have to be replaced,
- Substrates can be exposed to higher enzyme concentrations than with dissolved enzymes, speeding up reaction rates.